FDA Guidelines Ibuprofen – What Every User Must Know? | QuickDawa
You probably have a bottle of ibuprofen sitting in your medicine cabinet right now.
Almost everyone does.
Got a headache? Pop an ibuprofen.
Sore back after a long day at work? Ibuprofen again.
Period cramps making you miserable? Yep, ibuprofen to the rescue.
It feels like the most normal, harmless thing in the world, right?
But here is the thing.
Even though ibuprofen is available without a prescription, the FDA (Food and Drug Administration) has a whole set of guidelines around it.
These FDA guidelines ibuprofen rules cover everything from how much you can take to what the label must say to serious safety warnings that were updated just a few years ago.
And honestly, most people never read any of it.
That little bottle has more rules behind it than you might imagine.
Understanding these guidelines is not about scaring you.
It is about keeping you safe and helping you use this medicine the smart way.
At QuickDawa, we are all about making medicine easy to understand.
So let’s break down everything you need to know about the FDA guidelines for ibuprofen in simple, clear language.
No medical degree required.
Just a few minutes of your time.
💊 What Is Ibuprofen?
Ibuprofen is a nonsteroidal anti-inflammatory drug, commonly called an NSAID.
Its ibuprofen classification puts it in the same family as naproxen and aspirin.
It was first developed by a team at Boots Group in the UK and later became hugely popular in the United States.
Brand names include Advil and Motrin, though many generic versions exist.
Ibuprofen treats pain, fever, and inflammation caused by conditions like headaches, toothaches, arthritis, muscle aches, and menstrual cramps.
It works by blocking chemicals in your body called prostaglandins that cause swelling and pain signals.
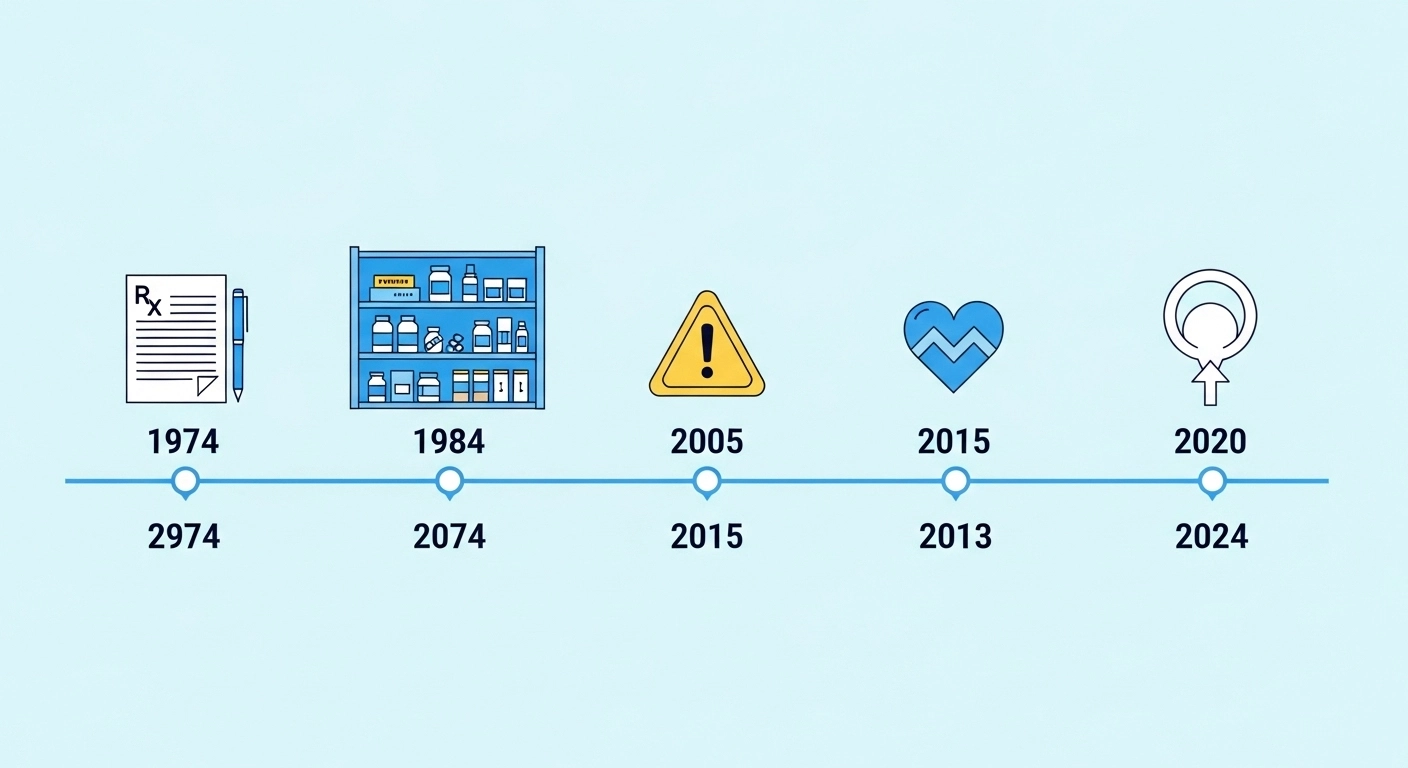
📜 The Ibuprofen History USA
The ibuprofen history USA story is actually pretty fascinating.
Let me walk you through the timeline.
The Early Days as a Prescription Drug
Ibuprofen was first approved by the FDA in 1974 as a prescription medication.
Back then, you needed a doctor’s note to get your hands on it.
It was mainly used to treat rheumatoid arthritis and other inflammatory conditions.
The prescription strength was typically 400 mg, 600 mg, or 800 mg per tablet.
Doctors loved it because it was effective and caused fewer stomach problems compared to aspirin at the time.
The Big OTC Switch in 1984
Here is where things got really interesting.
In 1984, the FDA approved ibuprofen for over-the-counter (OTC) sale at a lower dose of 200 mg.
This was a huge deal.
It was one of the first major prescription-to-OTC switches for a pain reliever in the US.
The ibuprofen OTC switch meant anyone could walk into a pharmacy or grocery store and buy it without a prescription.
Advil hit the shelves first, followed by Motrin IB shortly after.
Sales exploded almost immediately.
Within a year, ibuprofen became one of the most popular OTC pain relievers in America.
Why Did the FDA Allow This?
The FDA reviewed years of safety data and determined that ibuprofen at 200 mg doses was safe enough for people to use on their own.
The key was keeping the OTC dose lower than the prescription strength.
At 200 mg per tablet, the risks were considered manageable for most healthy adults when used as directed.
So is ibuprofen FDA approved? Absolutely, yes.
It has been FDA approved since 1974 for prescription use and since 1984 for OTC use.
🏛️ Current FDA Stance on NSAIDs (Including Ibuprofen)
Now let’s talk about where things stand today.
The FDA does not just approve a drug and forget about it.
They keep watching, reviewing new studies, and updating their guidelines.
And over the years, the FDA has become increasingly cautious about all NSAIDs, including ibuprofen.
The 2005 Warning
In 2005, the FDA first required NSAID labels to carry warnings about potential cardiovascular risks.
This came after another NSAID called Vioxx was pulled from the market due to heart attack and stroke risks.
The FDA decided that all NSAIDs needed stronger warnings, not just the ones that had been in trouble.
Ibuprofen was included in this.
The 2015 Strengthened Warning
Then in 2015, the FDA took an even bigger step.
They strengthened the existing heart attack and stroke warning on all prescription and OTC NSAIDs.
The updated warning stated that:
- The risk of heart attack or stroke can happen as early as the first weeks of using an NSAID.
- The risk increases with longer use.
- The risk is greater at higher doses.
- People with or without heart disease can be affected.
- People who already have heart disease are at even greater risk.
This was not a small update.
It was a major safety communication from the FDA that changed how millions of people should think about popping ibuprofen.
What This Means for You
The FDA is not saying you should never take ibuprofen.
They are saying you should use the lowest effective dose for the shortest time possible.
That is the golden rule right there.
If two days of ibuprofen fixes your headache problem, do not take it for two weeks.
🏷️ FDA Labeling Requirements
Every bottle of OTC ibuprofen you buy has a Drug Facts Label on it.
This label is not optional.
The FDA requires it by law, and it must follow a very specific format.
Let me break down what each section means and why it matters.
Active Ingredient
This section lists ibuprofen and the amount per tablet, usually 200 mg for OTC products.
It also identifies it as an NSAID so you know what type of drug you are taking.
Purpose
This tells you the drug’s category.
For ibuprofen, it says “Pain reliever/fever reducer.”
Simple and clear.
Uses
The label lists the specific conditions ibuprofen is approved to temporarily treat:
- Headache
- Toothache
- Backache
- Menstrual cramps
- Minor pain of arthritis
- Muscular aches
- Common cold symptoms
- Fever reduction
These are the FDA-approved uses for OTC ibuprofen.
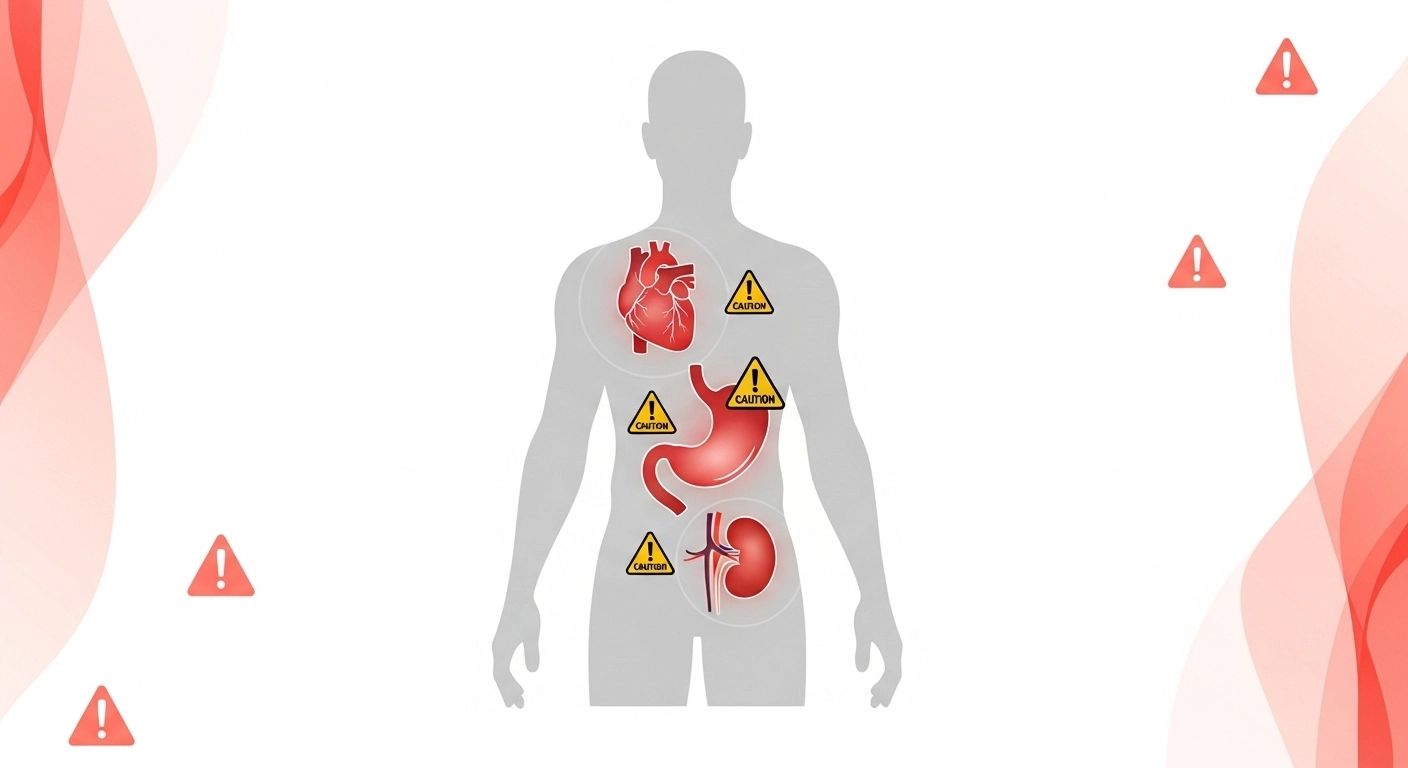
Warnings Section
This is the longest and most important part of the label.
The FDA requires several specific warnings:
Heart Attack and Stroke Warning:
“NSAIDs may cause an increased risk of serious cardiovascular thrombotic events, heart attack, and stroke, which can be fatal.”
Stomach Bleeding Warning:
“This product contains an NSAID, which may cause severe stomach bleeding.”
The risk is higher if you:
- Are age 60 or older
- Have a history of stomach ulcers or bleeding problems
- Take blood thinners or steroids
- Take other drugs containing NSAIDs
- Drink three or more alcoholic beverages daily
- Take more than directed or for a longer time than directed
Allergy Alert:
Ibuprofen may cause a severe allergic reaction, especially in people allergic to aspirin.
Symptoms include hives, facial swelling, asthma (wheezing), shock, and skin reddening.
Reye’s Syndrome Warning:
Children and teenagers who have or are recovering from chicken pox or flu should not use this product.
This is because NSAIDs have been linked to Reye’s syndrome, a rare but serious condition.
Directions
The FDA mandates specific dosing instructions on the label:
- Adults and children 12 years and older: Take 1 tablet every 4 to 6 hours while symptoms last.
- For the first dose, you may take 2 tablets within the first hour.
- Do not take more than 3 tablets in 24 hours unless directed by a doctor.
- Use the smallest effective dose.
- Children under 12: Ask a doctor before giving ibuprofen.
These directions are not suggestions.
The FDA requires them to be printed clearly on every OTC ibuprofen product.
Other Information
This section may include storage instructions and additional details like the amount of certain nutrients (such as potassium or sodium) per tablet.
Inactive Ingredients
Lists all non-medicinal ingredients like coatings, colorings, and fillers.
This matters for people with allergies to specific inactive ingredients.
At QuickDawa, we always encourage people to actually read these labels.
It takes 60 seconds and it could genuinely protect your health.
📢 Recent FDA Safety Communications About Ibuprofen
The FDA regularly publishes safety communications when new information comes to light about a drug.
Here are the most important ones related to ibuprofen and NSAIDs.
2015: Updated Cardiovascular Warning
As I mentioned earlier, this was the big one.
The FDA reviewed new safety information and concluded that the risk of heart attack and stroke with NSAIDs was more serious than previously thought.
They required all NSAID labels, both prescription and OTC, to be updated with stronger language.
2020: FDA Warning About NSAIDs in Pregnancy
In October 2020, the FDA issued a new safety communication requiring a label update for all NSAIDs regarding use during pregnancy.
The key point was that using NSAIDs, including ibuprofen, at 20 weeks or later in pregnancy can cause rare but serious kidney problems in the unborn baby.
This can lead to low amniotic fluid levels, which can create complications.
The FDA now recommends:
- Avoid ibuprofen at 20 weeks of pregnancy and beyond unless specifically told by your doctor.
- Between 20 and 30 weeks, use only if clearly necessary and for the shortest time.
- Do not use at all after 30 weeks of pregnancy.
This was a significant update that many people still do not know about.
2023 and Beyond: Ongoing Monitoring
The FDA continues to monitor NSAID safety data.
They review adverse event reports, new clinical studies, and real-world evidence.
If new risks emerge, they can require additional label changes, issue public warnings, or even restrict how the drug is sold.
This is exactly why staying informed about FDA guidelines ibuprofen rules matters.
The guidelines can change as science learns more.
🚨 When to Call Your Doctor
Ibuprofen is generally safe when used correctly.
But there are times when you absolutely need to call your doctor or seek emergency help.
Call your doctor right away if you experience:
- Chest pain, shortness of breath, or sudden weakness on one side of your body
- Slurred speech or sudden vision changes
- Black, bloody, or tarry stools
- Vomiting blood or material that looks like coffee grounds
- Severe stomach pain that does not go away
- Swelling of the face, throat, or tongue
- Skin rash, blisters, or peeling
- Unusual weight gain or swelling in the legs and feet
- Signs of kidney problems like changes in urine output
Also talk to your doctor before taking ibuprofen if you:
- Have heart disease, high blood pressure, or a history of stroke
- Have kidney or liver disease
- Have stomach ulcers or a bleeding disorder
- Are pregnant or planning to become pregnant
- Take blood thinners, steroids, or other NSAIDs
- Are over 60 years old
Do not take chances with your health.
When in doubt, ask a professional.
🙋 Frequently Asked Questions About Ibuprofen
Q: Can you take ibuprofen with acetaminophen at the same time?
A: Yes, many doctors actually recommend alternating or combining ibuprofen with acetaminophen (Tylenol) for better pain relief because they work differently in the body. However, always confirm with your doctor or pharmacist first.
Q: How long does ibuprofen take to work for pain relief?
A: Ibuprofen usually starts working within 20 to 30 minutes after you take it. Full effects are typically felt within 1 to 2 hours, and the relief generally lasts 4 to 6 hours.
Q: Is ibuprofen safe for children under 12?
A: The FDA says OTC adult ibuprofen tablets should not be given to children under 12 without a doctor’s guidance. However, children’s ibuprofen products (like Children’s Motrin or Children’s Advil) are specifically formulated with proper dosing for younger kids based on weight and age.
Q: Ibuprofen vs naproxen: which one is better for inflammation?
A: Both are NSAIDs that reduce inflammation. The main difference is that naproxen lasts longer (up to 12 hours vs 4 to 6 hours for ibuprofen), so you take fewer doses. But ibuprofen may work faster. The best choice depends on your specific situation, so ask your doctor.
Q: Is ibuprofen safe for toothache pain?
A: Yes, ibuprofen is one of the most commonly recommended OTC pain relievers for toothache pain. The American Dental Association often recommends ibuprofen combined with acetaminophen as a first-line treatment for dental pain. But if the toothache persists, see your dentist.
🌐 Sources and External References
Here are trusted sources where you can learn more:
- FDA Drug Label for Ibuprofen
- MedlinePlus: Ibuprofen
- Drugs.com: Ibuprofen Full Information
- WebMD: Ibuprofen Oral
- Mayo Clinic: Ibuprofen (Oral Route)
- FDA Safety Communication: NSAIDs and Pregnancy
🏁 Final Thoughts: Knowledge Is Your Best Medicine
Let’s quickly recap what we covered today.
Ibuprofen has been FDA approved since 1974 and moved to OTC status in 1984.
The FDA guidelines ibuprofen rules include strong warnings about heart attack, stroke, and stomach bleeding risks.
The Drug Facts Label on every bottle is your best friend, so actually read it.
Recent FDA updates have added new warnings about using ibuprofen during pregnancy after 20 weeks.
And the golden rule is always this: use the lowest dose for the shortest time that works for you.
At QuickDawa, we believe that understanding your medicine is just as important as taking it. Knowledge is the first step toward better health. If you found this guide helpful, share it with someone who needs it and explore more medicine guides on QuickDawa.com for trusted, easy-to-understand health information.
Got questions or want to share your experience with ibuprofen? Drop a comment below or reach out through our QuickDawa Contact Page. We love hearing from our readers.
⚠️ Medical Disclaimer
The information provided in this article is intended for educational and informational purposes only. It is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the guidance of your qualified doctor, pharmacist, or healthcare provider with any questions you may have regarding a medical condition, medication, or treatment plan.
Never disregard professional medical advice or delay seeking it because of something you read on QuickDawa.com or any other website. If you think you may have a medical emergency, call your doctor, go to the nearest emergency room, or call emergency services immediately.
Drug information, dosages, and interactions described in this post are provided for general awareness only and may not apply to your individual health situation. Medications affect people differently based on age, weight, health conditions, and other factors.
QuickDawa.com and its authors do not assume any liability for any consequences arising from the use or misuse of information presented on this website.